Accelerate formulation development, validate shelf-life and meet UK/EU regulations for pharmaceutical stability testing with LUM’s advanced analytical solutions, available only through Adaptive Instruments in the UK and Ireland.
Trusted by pharmaceutical, biotech and cosmetic formulation experts for rapid, reproducible stability insights.
Pharmaceutical stability testing underpins safe and effective medicines and cosmetics. However, some traditional methods of pharmaceutical shelf-life testing take months, rely on visual checks and manual scoring, and can make it difficult to diagnose instability mechanisms with confidence.
Adaptive Instruments delivers a more powerful approach: science-grade stability testing for drugs and cosmetics using LUM GmbH instruments. Available exclusively through us, LUM instruments dramatically reduce testing time, improve decision-making, and help QA,QC, R&D and academic teams generate robust, reproducible evidence.
LUM instruments allow scientists to observe separation, particle migration, structural changes and dispersion behaviour under controlled temperature, stress and acceleration.
Multi-Wavelength Dispersion Analyser
Get a fast read on the most stable formulations without waiting months from the widely-used accelerated stability analyser: the LUMiSizer.
Compliance: ISO 13318, ISO/TR 13097, ISO/TR 18811, ISO 18747
Multi-Wavelength Separation Analyser
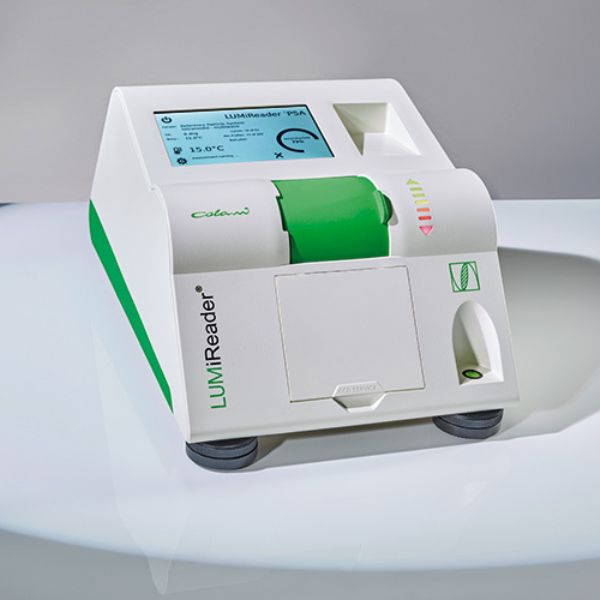
Accelerate stability measurement at the product’s original concentration.
Compliance: ISO/TR 18811, ISO 13317, ASTM D7827, ISO 18747, ISO/TR 13097
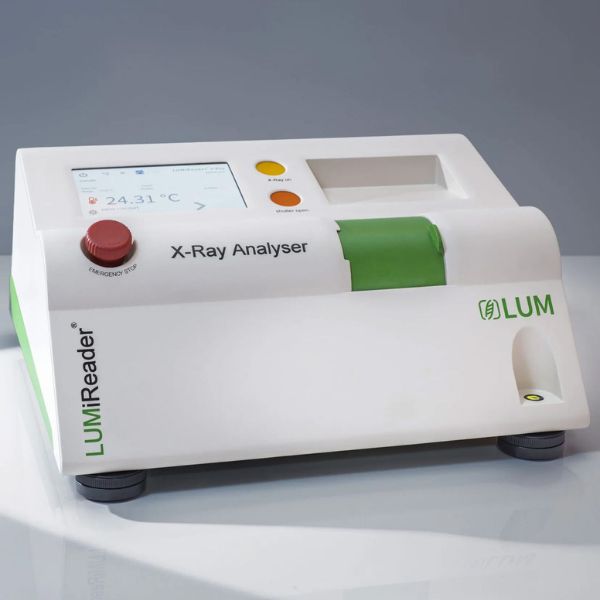
X-Ray Gravity Separation & Particle Analyser
Diagnose opaque products without disturbance over time.
Compliance: ISO 13317-3, ISO 18747, ISO/TR 13097, ISO/TR 18811
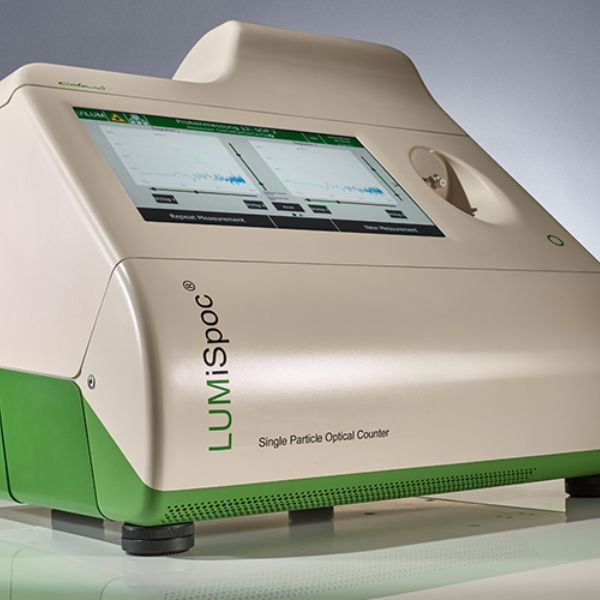
LUMiSpoc Single Particle Optical Counter
Count and size sub-micron to micron particles and track aggregation.
Compliance: ISO 21501-2
Pharmaceutical products, whether medicines, drug formulations, cosmetics or dermatological products, face common issues when it comes to stability testing:
We help scientists in QA, QC and R&D departments solve these problems:

Adaptive Instruments supports the pharmaceutical drugs and cosmetics industries by facilitating fast, comprehensive and compliant stability testing for:
Adaptive Instruments and LUM technology is widely used across the pharmaceutical landscape to support formulation development and quality assurance. Relevant product categories include:
For pharmaceutical-grade cosmetics, Adaptive Instruments and LUM technology supports fast, evidence-based stability assessment. Relevant products include:
Explore curated resources covering advanced stability testing methods, case studies, scientific evaluations and more below. Simply click the links, fill in the brief form and receive the resource straight to your inbox.

Stability Testing Solutions for Pharmaceuticals
Learn more about LUM’s analytical instruments for accelerated and real-time stability testing.

Stability Testing & Particle Analysis for Cosmetics
Learn how LUM’s analytical instruments accelerate stability testing and formulation screening.

Vaccine & Adjuvant Dispersion Stability
See how LUM’s STEP-Technology enables real-time particle characterisation of vaccines.

Tensile Strength Testing of Medical Tape & Plasters
See how the LUMiFrac Adhesion Analyser delivers precise multisample testing.
Contact our technical team for tailored advice on selecting the right LUM instrument for your application.